Bioinstrumentation & Medical Devices
Does Biomedical Engineering Involve A Lot Of Math?
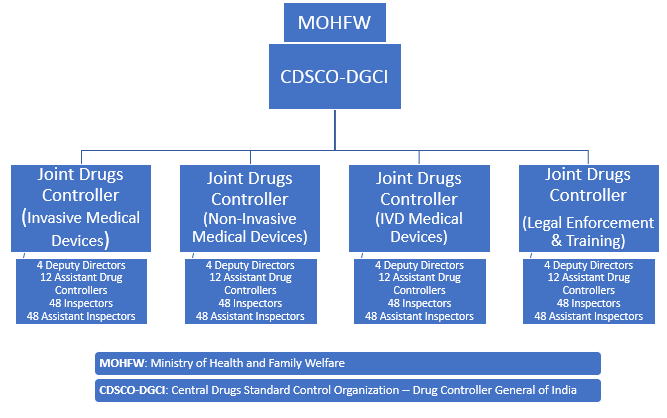
A Notified Body is a public or private organisation that has been accredited to validate the compliance of the gadget to the European Directive. Medical gadgets that pertain to class I (on situation they don’t require sterilization or do not measure a perform) could be marketed purely by self-certification. The regulatory authorities acknowledge different classes of medical gadgets based mostly on their potential for harm if misused, design complexity, and their use traits. The authorities additionally acknowledge that some devices are provided together with drugs, and regulation of those combination merchandise takes this factor into consideration. A international definition for medical system is troublesome to determine as a result of there are quite a few regulatory our bodies worldwide overseeing the advertising of medical units.
Often a portion of the definition of a medical device is intended to distinguish between medical devices and medicines, as the regulatory requirements of the 2 are completely different. Definitions also usually acknowledge In vitro diagnostics as a subclass of medical devices and establish equipment as medical devices.
After graduating, she or he may decide to concentrate on specific forms of gear used, typically in nuclear drugs, surgery, radiology, medical laboratory, dialysis, or intensive care. Class III medical units are those gadgets that have a high risk to the patient and/or person. These units often sustain or assist life, are implanted, or current potential unreasonable danger of illness or injury.
Examples of Class III gadgets embody implantable … Read More



